You must be a registered member to access this simulation. Registration is free and your privacy will be respected. If you are not already a member, click here to register. To reference this educational web simulation according to the APA style for Web references, use: |
Click to run simulation |
||||
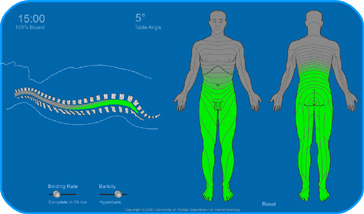
This transparent reality simulation implemented with Adobe Director uses a network of 6,934 cells to predict drug distribution in the spinal cavity using a heuristic modeling technique. It considers baricity, diffusion, binding rate, and the general shape and orientation of the spinal cavity. Binding rate is assumed to be linear. Time to complete binding may be adjusted from 10 to 30 minutes. Links to time-compressed movies of the simulation are listed below. Learning Objectives A hypobaric block will float, and a hyperbaric one will sink. The orientation of the patient is important. Carefully consider where the drug may distribute before moving the patient when the drug binding is not complete. The innervation of the diaphragm originates from C3-5 and the intercostals from the associated dermatomes. Patients with a hyperbaric spinal should not be laid prone before the drug is completely bound unless the neck is rigorously kept above the thoracic curve until drug binding is complete. Whatever drug distributes past the thoracic curve has a potential to sink into the cervical area and depress breathing. Patients with a hypobaric spinal should not be laid supine unless the drug is completely bound. Whatever distributes past the thoracic curve has a potential to float into the cervical area and depress breathing. Based on spine curvature, at the leading edge of the drug distribution zone, drug penetration may differ by up to two vertebrae at the dorsal and ventral sides, depending on patient position. Simulation Screen Capture Videos The spinal anesthesia simulation works in real time and is therefore slow. These time-compressed movies have been made from screen captures of the simulation. They typically last about 3 to 4 minutes. Modeling The heuristic model is based on a network of 6,934 cells. Each cell corresponds to one pixel and contains references to its direct neighboring cells (maximum of 4). The cells are assigned a quantity, analogous to a drug, and pass a fraction of this quantity to their direct neighbors. The diffusion rate between the cells is regulated by allowing each cell to exchange only a fraction of their quantity. Drug binding is modeled by allowing each cell to permanently withhold a fraction from further exchange. At each time step, each cell takes its respective drug quantity, subtracts a fraction of it, and considers this fraction bound. In the case of an isobaric drug, the cell then takes what is left and exchanges it equally with its direct neighbors. In the case of a hyperbaric or hypobaric drug, the exchange is biased straight down or straight up respectively, independent of patient orientation. Baricity is modeled by the strength of this bias. After every cell has passed the prescribed quantities to its direct neighbors, all cells recalculate their quantities. The cells then set their respective pixel to a color that is scaled to their free and bound quantities. Cells related to a dermatome copy their color to the corresponding portion of the dermatome charts. Diffusion and gravity effects can be tuned so that the resulting drug distribution resembles what is described in textbooks and by clinicians. The simulation clock runs slightly slower than real time due to current limitations in presenting such a model on the web, i.e. in a virtual machine encapsulated in a browser. Time-compressed demonstration videos covering relevant teaching points are posted above. Bernards CM: Epidural and Spinal Anesthesia in Barash PG, Cullen BF, Stoelting RK (eds.): Clinical Anesthesia (5th ed.), Lippincott, Williams & Wilkins, Philadelphia, 2006 |
|||||